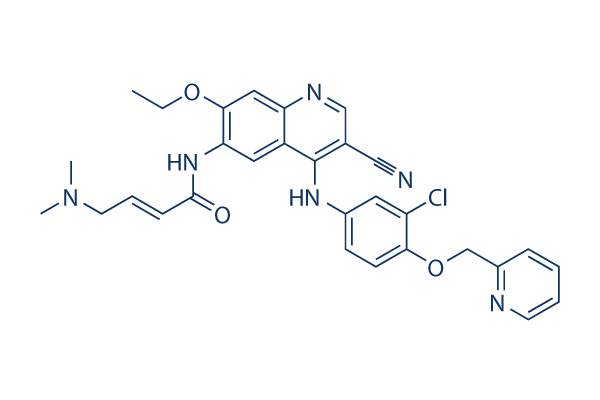
Pint-Pharma - Puma Biotechnology and Pint Pharma Enter into Exclusive Licensing Agreement to Commercialize NERLYNX® (neratinib) in Latin America

Puma Biotechnology Presents Outcomes from the Metastatic Breast Cancer Cohort of the SUMMIT Trial at the ASCO 2022 Annual Meeting
Puma Biotechnology Presents Final Overall Survival Analysis from the Phase III ExteNET Trial at the 2020 SABCS | Business Wire
Puma Biotechnology: Waiting For Commercial Numbers To Catch Up To Clinical Data (NASDAQ:PBYI) | Seeking Alpha

Cancer hopeful neratinib plagued by diarrhea, analysis shows, but Puma cites a solution | Fierce Pharma
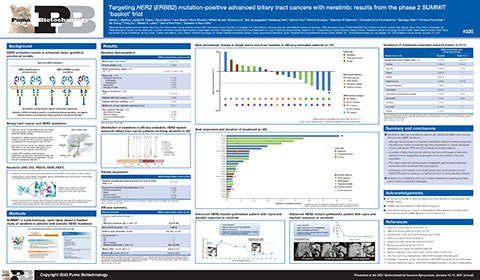
Puma Biotechnology Presents Interim Results from the Biliary Tract Cancers Cohort of the Phase II SUMMIT “Basket” Trial of Neratinib at ASCO GI | Business Wire
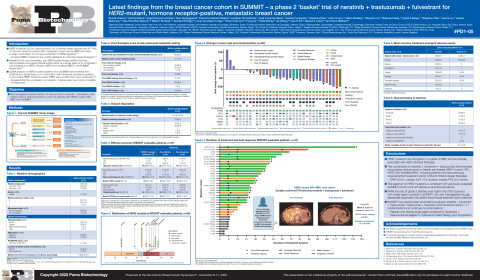
Puma Biotechnology Presents Updated Results from the Phase II SUMMIT Trial of Neratinib for HER2-Mutant, HR-Positive Metastatic Breast Cancer at SABCS 2020 | Business Wire

Puma Biotechnology Receives US FDA Approval of Supplemental New Drug Application for Neratinib to Treat HER2-Positive Metastatic Breast Cancer

PUMA BIOTECHNOLOGY: Announces U.S. FDA Acceptance of Supplemental New Drug Application for Neratinib to Treat HER2-Positive Metastatic Breast Cancer | FDA Health News

Puma Biotechnology, Inc. - Puma Biotechnology Presents Interim Results of Phase II CONTROL Trial of Neratinib in Extended Adjuvant Treatment of HER2-Positive Early Stage Breast Cancer at the 2019 SABCS
Dose escalation for mitigating diarrhea: Ranked tolerability assessment of antidiarrheal regimens in patients receiving neratin